LETTER
Why do Pain Physicians Not Routinely Use Mixed Opioids for the Prevention of Neuraxial Opioid-induced Pruritus?
Borja Mugabure Bujedo*
Article Information
Identifiers and Pagination:
Year: 2017Volume: 10
First Page: 14
Last Page: 21
Publisher ID: TOPAINJ-10-14
DOI: 10.2174/1876386301710010014
Article History:
Received Date: 20/12/2016Revision Received Date: 14/02/2017
Acceptance Date: 17/02/2017
Electronic publication date: 31/05/2017
Collection year: 2017

open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: (https://creativecommons.org/licenses/by/4.0/legalcode). This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background:
Pruritus is a very disturbing secondary effect that appears after epidural or intrathecal administration of opioid drugs, especially in the management of postoperative pain. It is induced by the activation of mu opioid receptors and it can often be even more unpleasant than the pain being treated.
Objective:
A wide variety of drugs with different mechanisms of action have been used, aiming at the prevention of pruritus, with varying results. The aim of this comprehensive review letter is to summarize the current evidence of the available pharmacological options to either treat or prevent pruritus induced by spinal opioids.
Method:
The articles used in the review were found through a search in Medline, PubMed and Cochrane Library up to December 2016, using the keywords “Neuraxial opioids”, “Intrathecal morphine”, “Pruritus”, “Naloxone”, “Nalbuphine” and “Butorphanol”.
Results:
The most useful drugs act on the mu and kappa opioid receptors. They are either mu opioid antagonists, like intravenous naloxone, or mixed opioids mu antagonists/kappa agonists, such as intravenous nalbuphine and intravenous or epidural butorphanol, the latter being able also for maintaining the analgesia.
Conclusion:
Both pruritus prevention and treatment remain a challenge in the treatment of patients receiving spinal opioids for postoperative pain. Recent findings suggest that mixed opioids must be added to evidence-based clinical guidelines for the management of pruritus induced by spinal opioids.
INTRODUCTION
Opioids analgesics are the most common analgesic drugs used for the management of severe pain. Opioids have always been considered the best option for the treatment of severe postoperative pain due to their high efficacy and good tolerability. Recent evidence-based clinical guidelines recommend a multimodal approach, using a neuraxial mixture of a strong opioid plus a local anesthetic, to improve the global analgesic effect, minimize adverse effects and to improve the overall patient satisfaction after major surgery [1]. However, a spinal opioid administration does not guarantee segmental analgesia in the spine without systemic adverse effects such as nausea, pruritus, urine retention and respiratory depression. The reason for these undesirable secondary effects could either be partial reuptake to the systemic blood circulation by epidural vessels or drug clearance from cerebrospinal fluid (CSF). In both cases, opioids reach brain receptors and produce centrally mediated analgesia [2]. Published evidence from clinical studies indicates that opioid bioavailability in the spinal cord biophase (percentage of an opioid drug that joins specific receptors on Rexed lamina II in the dorsal horn) is negatively correlated with liposolubility. Bioavailability is therefore higher for hydrophilic opioids like morphine than for lipophilic ones like fentanyl and sufentanil [3]. It has been observed that less than 5% of sulfate morphine, which is epidurally administrated as a bolus, could reach this biophase. The relationship between the analgesic effect of intravenous and intrathecal morphine is 1/200, so that 100-mcg intrathecal morphine corresponds with 20 mg given intravenously. These lower doses and more targeted delivery make spinal opioid administration an attractive way of improving postoperative pain scores [4].
Pruritus is a very disturbing secondary effect that appears after epidural or intrathecal administration of opioid drugs especially in the management of postoperative pain. It is induced by the activation of mu opioid receptors and it can often be even more unpleasant that the pain being treated. The overall incidence of pruritus ranges from 40.6% to 90%. The incidence of pruritus after spinal opioid administration is higher in postpartum patients (83%) than in non-pregnant patients (69%) including males and females [5]. Pruritus begins shortly after opioid-mediated analgesia with the onset depending on the type, route, and dosage of opioid used. Pruritus invoked by lipid-soluble drugs such as sufentanil and fentanyl presents shorter duration and the addition of local anesthetics and the use of the minimum effective dose seems to decrease the prevalence of itching. On the other hand, pruritus invoked by intrathecal morphine is of longer duration and is hard to treat. Pruritus, start on the trunk, nose, around the eyes and is usually located in areas of the face innervated by the trigeminal nerve. Both pruritus prevention and treatment remain a challenge in the management of patients receiving spinal opioids for postoperative pain. A wide variety of drugs with different mechanisms of action have been used with widely varying results and specially focused on its prevention (Fig. 1) [6].
 |
Fig. (1). Most common used drugs in the management of opioid-induced pruritus. |
DISCUSSION
Pruritus occurs in patients with very different underlying diseases as it is caused by various pathologic mechanisms. It can be present in patients suffering from uremic pruritus, cholestatic pruritus, HIV-associated pruritus and opioid-induced pruritus. In palliative care patients, pruritus is not the most prevalent but is one of the most puzzling side effects of opioid use. It can cause considerable discomfort and affects patients’ quality of life. In a recent Cochrane Review published in 2016 (50 studies, n=1916 patients) there was not enough evidence to indicate any possibly useful treatment for particular causes of pruritus. These included gabapentin, nalfurafine and cromolyn sodium for itch associated with chronic kidney disease, and rifampicin and flumecinol for pruritus related to liver problems. Paroxetine may be useful for palliative care patients although evidence was only available from one study. Overall, most of the drugs caused mild side effects. Moreover, naltrexone showed by far the most side effects, and overall evidence ranged from very low to moderate quality [7].
The primary mechanism of neuraxial opioid-induced pruritus is unclear. To date, many mechanisms have been postulated and include the presence of “Trigeminal itch center” in the central nervous system, the medullary dorsal horn activation and antagonism of inhibitory transmitters, the modulation of the serotonergic pathway, and finally, the theory that links pain and pruritus. It seems that pain and pruritus are transmitted by the same sensory neurons, namely small unmyelinated nerve fibers (C-fibers), and the consequent release of prostaglandins (PGE1/ PGE2) enhances C-fiber transmission to the central nervous system, which potentiates pruritus [8]. Moreover, it has also been demonstrated in animal studies that activation of mu opioid receptors (MOR) induces pruritus, but MOR antagonist and kappa opioid receptors (KOR) agonist decreases this unpleasant scratching feeling. In this review letter, we discuss the need to include mixed opioids, kappa agonists/ μ antagonist like butorphanol and nalbuphine in evidence-based practical clinical guidelines [9].
1. OPIOID RECEPTOR ANTAGONISTS
MOR is either responsible for pain modulation or some side effects, like pruritus, nausea and vomiting or respiratory depression. Therefore, MOR antagonist drugs should be the first line for all spinal opioid-induced side effects management, but a narrow clinical range is observed to maintain residual analgesia [6, 8, 9].
Nalmefene is a MOR antagonist drug developed in the seventies that has mainly been used for the treatment of opioid overdose and alcoholism. It also has good results for the treatment of established pruritus induced by neuraxial opioids in primates. Ko et al. demonstrated as a single prophylactic dose of 32 μg/kg nalmefene were very effective in reducing pruritus but also intrathecal morphine-induced analgesia. It developed an apparent deviation to the right of the dose/response curve in primates studied against both clinical parameters. This fact demonstrated that the therapeutic window is close to the production of analgesia and pruritus, both mediated by MOR [10]. Clocinamox, a selective MOR antagonist pre-treatment in an animal model, inhibited scratching induced by spinal opioids in primates, but the KOR antagonist binaltorfimina or the delta opioid receptor (DOR) antagonist naltrindol did not produce this clinical effect. This fact would explain the great antipruritic role of MOR antagonists [11]. Accordingly, some studies have evaluated the efficacy of naloxone, naltrexone and methylnaltrexone in the prevention of pruritus, but results observed were variable. A small dose, intravenously or in infusion, naloxone has the greatest evidence for pruritus prevention in the adult patient. Moreover, a continuous infusion is expected to produce less fluctuation of concentrations than a bolus injection and compensates for the short half-life of naloxone [12]. The results showed how an intravenous dose of 0.25 to 1 mcg/ kg / h was the most efficient dose range without affecting the analgesia. An excellent systematic review including 834 patients [13] concluded that the intravenous naloxone was an active drug in the treatment of either pruritus or nausea induced by spinal opioids, without increasing the pain score. Higher doses at 2 mcg/ kg / h were more likely to lead to the reversal of the analgesia and therefore should not be widely used in clinical practice. Another meta-analysis [14] about the efficacy of intravenous naloxone, either as a continuous infusion or patient-controlled analgesia, for postoperative pruritus, revealed that naloxone was associated with a significant decrease in pruritus or nausea without any increase in pain scores. However, a review from the University of York criticizes these results. Their DARE reviews system (Database of Abstracts of Reviews of Effects) states that due to the heterogeneity of the studies included in this systematic review, the high percentage of methodological limitations and the poor evidence base suggest that these conclusions about naloxone efficacy may not be reliable.
Moreover, another recent meta-analysis published in 2016 demonstrated the effectiveness of naloxone in the treatment of the opioid drugs adverse effects. 30 studies including 1138 patients were included in this meta-analysis. An exhaustive pooled analysis concluded that naloxone significantly reduced the occurrence of either pruritus (risk ratio RR = 0.252, 95%, confidence interval CI = 0.137-0.464) or vomiting (RR = 0.338, 95% CI = 0.192-0.593). However, naloxone did not relieve pain (standardized mean difference SMD = -0.052, 95% CI = -0.453 to 0.348) and somnolence (RR = 0.561, 95% CI = 0.287 to 1.097). Additionally, there was no significant publication bias between the included studies (Begg test, P = 0.602; Egger test, P = 0.388). After a careful selection, only six studies, which examined the effects of naloxone in opioid-induced pruritus, were included. The spinal route was used in four of them, and the systemic one in the two lasted studies. The analysis finally concluded that there was a high and significant heterogeneity between the included studies (I2 Index=60.3%, p=0.027), so the meta-analysis was based on the random effects model, so the data must be observed with caution. The results indicated that the incidence pruritus rate was significantly lower in the naloxone group versus the control group. The author´s conclusion was that naloxone might act as an effective strategy for either treatment or prophylaxis of opioid-induced pruritus, nausea, and vomiting in clinical practice [15].
However, under the conditions of a recent study, a single dose of 12 mg subcutaneous methylnaltrexone did not reduce the overall pruritus severity among an obstetric population after receiving 100 mcg of intrathecal morphine plus fentanyl 15 mcg for cesarean delivery. Prophylactic treatment with a peripheral mu antagonist was ineffective for the treatment of intrathecal morphine-induced pruritus, although a small clinical effect should not be excluded [16]. Similar results were found in an orthopedic surgery study. In this study, subcutaneous administration of methylnaltrexone was not effective in reducing postoperative urinary retention and pruritus, but it reduced the incidence of either nausea or vomiting after intrathecal bupivacaine and morphine [17].
2. MIXED OPIOID RECEPTOR AGONIST/ANTAGONIST
Mixed opioid KOR agonists / MOR antagonist such as nalbuphine or partial agonists as butorphanol and pentazocine have a great potential to mitigate the adverse effects and to improve the analgesic effects of opioids. The experimental studies on macaccus rhesus have demonstrated that both MOR antagonists and KOR agonists are useful in relieving itching induced by the intrathecal morphine-induced in primates [18]. It is logical to propose to translate these good results to human clinical practice if the available current literature on animal models demonstrates this fact.
2.1. Pentazocine and Butorphanol
Pentazocine’s and butorphanol’s effectiveness has been studied in the management of opioid-induced pruritus with positive results [19]. A randomized trial that investigates the effectiveness of pentazocine for the treatment of pruritus associated with the intrathecal injection of morphine concluded that 15 mg intravenous pentazocine was superior to 4 mg intravenous ondansetron. Pentazocine also had a lower pruritus recurrence rate [20]. A recent systematic review on the effectiveness of butorphanol included 16 trials, and 795 patients were analyzed [21]. Butorphanol reduced opioid-induced pruritus either by intravenous or epidural routes with an RR of 0.22 (95% CI 0.10 to 0.45) and RR 0.24 (95% CI 0.16 to 0.36), respectively. When using epidural butorphanol, the number of patients seeking itching rescue treatment was reduced (RR 0.57; 95% CI 0.41 to 0.81). Butorphanol also diminished the intensity of postoperative pain at four, eight and 12 hours, as were measured VAS score (visual analogue scale) differences of -0.29 (95% CI -0.52 to -0.05), -0.30 (95% CI -0.56 to -0.04) and -0.23 (95% CI -0.46 to -0.01). However, epidural but not intravenous butorphanol reduced postoperative nausea and vomiting (PONV) (RR 0.35; 95% CI: 0.19 to 0.66). Moreover, butorphanol did not increase respiratory depression (RR 0.71; 95% CI 0.31 to 1.63), dizziness (RR 2.45; 95% CI 0.35 to 17.14) or drowsiness (RR 0.71; 95% CI 0.22 to 2.37). The authors´ conclusion was that butorphanol administered with morphine might be an effective strategy to prevent itching due to its effect of reducing the intensity of the pain and PONV without increasing other side effects during the perioperative period.
In another study not included in the last meta-analysis, it was demonstrated that continuous epidural bupivacaine 0.1% plus butorphanol 0.004% at a rate of 2 ml/h, in patients undergoing elective cesarean section, decreased either incidence or severity of 100-mcg intrathecal morphine-related pruritus without adversely affecting the quality of postoperative analgesia. The impact of pruritus at 48 h was significantly lower in the butorphanol group (16.3 vs. 52.5%; p< 0.001). Furthermore, compared with the saline group, the pruritus severity was also more moderate with epidural butorphanol at 3, 6 and 9 h (p < 0.008) [22].
2.2. Nalbuphine
Nalbuphine hydrochloride is a potent opioid analgesic essentially equivalent to that of morphine on a milligram basis. Recent studies show that Nalbuphine binds to mu, kappa, and delta receptors, but not to sigma receptors so it can be defined as primarily kappa agonist/partial mu antagonist analgesic. Nalbuphine onset of action occurs within 2 minutes after intravenous administration, and in less than 15 minutes following subcutaneous or intramuscular injection. The plasma half-life is 5 hours, and in clinical studies, analgesic activity has been reported to range from 3 to 6 hours. Nalbuphine is ten times as potent as pentazocine referring to opioid antagonist activity. Nalbuphine may produce the same degree of respiratory depression as equianalgesic doses of morphine. Moreover, Nalbuphine exhibits a ceiling clinical effect such that increases in a dose greater than 30 mg do not produce further respiratory depression, always in the absence of other CNS active medications affecting patient respiration [23].
In a 2016 systematic review on nalbuphine [24], 1129 patients among ten studies met all the inclusion criteria. Among them, nine of them were randomized controlled trials and one a clinical case report. Pruritus incidence was higher in the group of patients who received spinal opioids versus those in the intravenous group. Nalbuphine provided the greatest effectiveness in the treatment of opioid-induced pruritus, compared with control, placebo or pharmacological agents such as naloxone, diphenhydramine, and propofol. There was no attenuation of the analgesia or increase of the sedation with small doses defined as 25% to 50% of the dose to treat the pain intravenously (2.5 to 5 mg versus 10 mg). This drug was also associated with a reduction of PONV and respiratory depression. The author´s conclusion was that nalbuphine is the best option to treat opioid-induced itching in patients receiving neuraxial opioids for acute pain related to childbirth or surgery. The author also recommended that nalbuphine should be used as a first-line treatment for opioid-induced pruritus.
Therefore, why do pain physicians not routinely use mixed opioids like nalbuphine for the prevention of neuraxial opioid-induced pruritus?
Maybe the essential fact is that there is a general lack of well-designed randomized trials in most common fields of surgery. Moreover, Januzzi RG explained the lack of transfer of these findings to the clinical practice due to other several factors:
- The absence of a clinical guidelines based on the evidence in this field.
- The counter-intuitive fact of dealing with the adverse effects of an opioid with another opioid drug.
- The off-label use of nalbuphine for the treatment of pruritus.
- The higher price of nalbuphine compared to other alternatives in the North American market.
There are a few economic implications with this new evidence that need consideration [24]. The cost of nalbuphine is substantially higher than the other commonly used antipruritic agents, such as diphenhydramine, but it is much lower than naloxone and propofol. For instance, a vial of IV nalbuphine (20 mg/mL) can cost approximately three times more than an IV diphenhydramine (50 mg/mL), $6.24 versus $1.90, at an average wholesale pricing. In contrast, intermittent IV nalbuphine is very economical compared with utilizing naloxone infusions, as the cost of naloxone alone is substantially higher at $18.90 per single dose. This fact is due to the associated cost of the IV administration set, saline solution, and infusion pumps, nursing labor to set up and monitor the patient, and finally titrate infusion. For those patients receiving mu-opioids drugs, treatment of pruritus with nalbuphine may increase their satisfaction with overall hospital care an outcome of interest to the third party payers and health care administrators. Low-dose nalbuphine is less sedating when compared with other antipruritic pharmacologic agents, such as ondansetron, diphenhydramine, and propofol. By eliminating or minimizing the use of these more sedating agents, patients are more likely to be awake and more able to participate in rehabilitation; and most importantly, the risk of respiratory depression associated with the additive sedation side effect of opioids, when combined with antihistamines and antiemetics, is decreased. Therefore, nalbuphine may facilitate timely discharge to home and prevent incurring the high cost associated with severe adverse outcomes, such as critical care admissions, death, or both.
The label text from NALBUPHINE® describes clinical use of this drug as follows (revised on September 2016) [25]: “Nalbuphine by itself has potent opioid antagonist activity at doses equal to or lower than its analgesic dose. When administered following or concurrent with mu agonist opioid analgesics (e.g., fentanyl, oxymorphone, morphine), nalbuphine could partially reverse or block opioid-induced respiratory depression from the mu agonist analgesic. Nalbuphine hydrochloride might precipitate withdrawal in patients dependent on opioid drugs. Therefore, it should be used with caution in patients who have been receiving mu opioid analgesics on a regular basis.”
As a consequence, Nalbuphine injection by intramuscular, subcutaneous or intravenous is only indicated for the management of moderate to severe pain enough to require an opioid analgesic and for which alternative treatments are inadequate. This opioid can also be used as a supplement to intraoperative balanced anesthesia, for postoperative analgesia, and for analgesia during labor and delivery in obstetrical patients older than 18 years old. Therefore, routine use of nalbuphine for opioid-induced pruritus in the postoperative setting is an “off-label” use and contra-intuitive in clinical practice. In a recent systematic Cochrane review about nalbuphine for the management of pre and postoperative pain in children, the overall quality of available evidence was weak, so could not definitively show that the analgesic efficacy of nalbuphine was superior compared to placebo. Therefore, this drug is not widely used in pediatric anesthesia except for sedation during diagnostic procedures, because of a ceiling effect that does not cause a respiratory depression [26].
With nalbuphine as the first-line treatment of opioid-induced pruritus, studies are needed to determine the dosing regimen that provides the maximal reduction of pruritus without affecting analgesia or increasing sedation. Recently, in a dosing study with n=90 parturient with intrathecal morphine after cesarean section, nalbuphine 2, 3 and 4 mg were compared regarding treatment efficacy, with treatment success rates at 87%, 97% and 100%, respectively. No significant differences between groups were observed in the percentage of success, or in adverse effects. However, pain score was significantly increased (P=0.004) with a nalbuphine dose at 4 mg. The authors subsequently recommended an optimal nalbuphine dose of 2 to 3 mg. Moreover, either intravenous nalbuphine (4 mg) or ondansetron (4 mg) was more effective than placebo for the prevention of pruritus. Indeed, nalbuphine was preferred to ondansetron because it was not excreted in the breast milk [27]. When the intramuscular route is used, 10 mg prophylactic nalbuphine was effective in decreasing the incidence and severity of pruritus and did not affect analgesia after cesarean surgery [28]. In this study, Nalbuphine proved better than diphenhydramine for prevention of epidural morphine-induced pruritus in patients who underwent cesarean section.
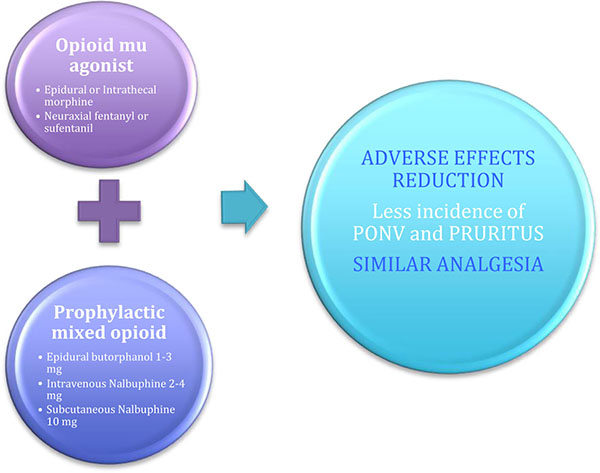 |
Fig. (2). Prevention strategy of opioid-induced pruritus mixing MOR opioids with agonist-antagonist MOR/KOR opioids. |
CONCLUSION
A large variety of drugs have been evaluated for the treatment of adverse effects induced by neuraxial opioids administration in the management of postoperative pain. Most of them have been discussed in recent years in systematic reviews, clinical trials or case reports articles, but results have been inconclusive. Since these drugs have not been enough studied in non-human primates, there is no translation from the results to evidence based clinical guidelines. These pharmacological studies suggest that Rhesus monkeys can and must serve as a surrogate species for human beings in preclinical studies to identify effective treatments for opioid-induced pruritus prevention. MOR antagonists like naloxone are the most effective drugs to reverse either pruritus or other secondary effects such as PONV and respiratory depression, after spinal opioids administration. However, a narrow action range is described for this group of drugs, and they can also reverse the analgesic effect. Despite this, the accumulated pharmacological experimental and recent clinical published evidence suggests the use of MOR antagonists and mixed mu and kappa partial agonists drugs, as the most efficient treatment for spinal opioid induced pruritus while maintaining analgesia. Moreover, partial or mixed agonist/antagonist opioids like intravenous or epidural butorphanol (1-3 mg) and nalbuphine (2-4 mg iv) have demonstrated the greatest effectiveness in the prevention and treatment and should be considered as the first-line treatment of pruritus induced by the neuraxial opioid administration (Fig. 2) .
DISCLOSURE
Part of this article has been previously published in Journal of Anesthesia & Critical Care: Open Access. Volume 6 Issue 2 - 2016.
CONFLICT OF INTEREST
The author confirms that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
The author is indebted to Dr. Brynjar Karlsson for reading this paper in an advanced draft form and making many useful suggestions for improving its content and style.
REFERENCES
| [1] | Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: A clinical practice guideline from the American Pain Society, the American society of regional anesthesia and pain medicine, and the American society of anesthesiologists committee on regional anesthesia, executive committee, and administrative council. J Pain 2016; 17(2): 131-57. |
| [2] | Bujedo BM, Santos SG, Azpiazu AU. A review of epidural and intrathecal opioids used in the management of postoperative pain. J Opioid Manag 2012; 8(3): 177-92. |
| [3] | Bujedo BM. Current evidence for spinal opioid selection in postoperative pain. Korean J Pain 2014; 27(3): 200-9. |
| [4] | Bujedo BM. Spinal opioid bioavailability in postoperative pain. Pain Pract 2014; 14(4): 350-64. |
| [5] | Bonnet MP, Mignon A, Mazoit JX, Ozier Y, Marret E. Analgesic efficacy and adverse effects of epidural morphine compared to parenteral opioids after elective caesarean section: A systematic review. Eur J Pain 2010; 14(9): 894.e1-9. |
| [6] | Bujedo BM. An Update on neuraxial opioid-Induced pruritus prevention. J Anesth Crit Care Open Access 2016; 6(2): 00226. [http://medcraveonline.com/JACCOA/JACCOA-06-00226.php]. |
| [7] | Siemens W, Xander C, Meerpohl JJ, et al. Pharmacological interventions for pruritus in adult palliative care patients. Cochrane Database Syst Rev 2016; 16(11): 00226. |
| [8] | Kumar K, Singh SI. Neuraxial opioid-induced pruritus: An update. J Anaesthesiol Clin Pharmacol 2013; 29(3): 303-7. |
| [9] | Ko MC. Neuraxial opioid-induced itch and its pharmacological antagonism. Handbook Exp Pharmacol 2015; 226: 315-35. |
| [10] | Ko MC, Naughton NN. An experimental itch model in monkeys: Characterization of intrathecal morphine-induced scratching and antinociception. Anesthesiology 2000; 92(3): 795-805. |
| [11] | Ko MC, Song MS, Edwards T, Lee H, Naughton NN. The role of central mu opioid receptors in opioid-induced itch in primates. J Pharmacol Exp Ther 2004; 310(1): 169-76. |
| [12] | Miller JL, Hagemann TM. Use of pure opioid antagonists for management of opioid-induced pruritus. Am J Health Syst Pharm 2011; 68(15): 1419-25. |
| [13] | Kjellberg F, Tramèr MR. Pharmacological control of opioid-induced pruritus: a quantitative systematic review of randomized trials. Eur J Anaesthesiol 2001; 18(6): 346-57. |
| [14] | Murphy JD, Gelfand HJ, Bicket MC, et al. Analgesic efficacy of intravenous naloxone for the treatment of postoperative pruritus: A meta-analysis. J Opioid Manag 2011; 7(4): 321-7. |
| [15] | He F, Jiang Y, Li L. The effect of naloxone treatment on opioid-induced side effects: A meta-analysis of randomized and controlled trails. Medicine (Baltimore) 2016; 95(37): e4729. |
| [16] | Paech M, Sng B, Ng L, Nathan E, Sia A, Carvalho B. Methylnaltrexone to prevent intrathecal morphine-induced pruritus after Caesarean delivery: A multicentre, randomized clinical trial. Br J Anaesth 2015; 114(3): 469-76. |
| [17] | Zand F, Amini A, Asadi S, Farbood A. The effect of methylnaltrexone on the side effects of intrathecal morphine after orthopedic surgery under spinal anesthesia. Pain Pract 2015; 15(4): 348-54. |
| [18] | Ko MC, Lee H, Song MS, et al. Activation of kappa-opioid receptors inhibits pruritus evoked by subcutaneous or intrathecal administration of morphine in monkeys. J Pharmacol Exp Ther 2003; 305(1): 173-9. |
| [19] | Dominguez JE, Habib AS. Prophylaxis and treatment of the side-effects of neuraxial morphine analgesia following cesarean delivery. Curr Opin Anaesthesiol 2013; 26(3): 288-95. |
| [20] | Tamdee D, Charuluxananan S, Punjasawadwong Y, Tawichasri C, Patumanond J, Sriprajittichai P. A randomized controlled trial of pentazocine versus ondansetron for the treatment of intrathecal morphine-induced pruritus in patients undergoing cesarean delivery. Anesth Analg 2009; 109(5): 1606-11. |
| [21] | Du BX, Song ZM, Wang K, et al. Butorphanol prevents morphine-induced pruritus without increasing pain and other side effects: A systematic review of randomized controlled trials. Can J Anaesth 2013; 60(9): 907-17. |
| [22] | Wu Z, Kong M, Chen J, Wen L, Wang J, Tan J. Continous epidural butorphanol decreases the incidence of intrathecal morphine-related pruritus after cesarean section: A randomized, double-blinded, placebo-controlled trial: Epidural butorphanol decreases the incidence of intrathecal morphine-related pruritus. Cell Biochem Biophys 2014; 70(1): 209-13. |
| [23] | Bullingham RE, McQuay HJ, Moore RA. Clinical pharmacokinetics of narcotic agonist-antagonist drugs. Clin Pharmacokinet 1983; 8(4): 332-43. |
| [24] | Jannuzzi RG. Nalbuphine for treatment of opioid-induced pruritus: A systematic review of literature. Clin J Pain 2016; 32(1): 87-93. |
| [25] | https://www.drugs.com/pro/nalbuphine.html. Available on the internet. Accessed on December 15th, 2016. |
| [26] | Schnabel A, Reichl SU, Zahn PK, Pogatzki-Zahn E. Nalbuphine for postoperative pain treatment in children. Cochrane Database Syst Rev 2014; 31(7): CD009583. |
| [27] | Moustafa AA, Baaror AS, Abdelazim IA. Comparative study between nalbuphine and ondansetron in prevention of intrathecal morphine-induced pruritus in women undergoing cesarean section. Anesth Essays Res 2016; 10(2): 238-44. |
| [28] | Liao CC, Chang CS, Tseng CH, et al. Efficacy of intramuscular nalbuphine versus diphenhydramine for the prevention of epidural morphine-induced pruritus after cesarean delivery. Chang Gung Med J 2011; 34: 172-8. [http://memo.cgu.edu.tw/cgmj 3402/340206.pdf]. |