All published articles of this journal are available on ScienceDirect.
A Systematic Review with Meta-analysis of Studies Comparing Response to Experimentally-evoked Pain Between Obese and Non-Obese Individuals
Abstract
Background:
The relationship between obesity and pain remains unclear. The aim of this systematic review was to determine whether response to experimentally-evoked pain differed between obese and non-obese individuals. Studies that compared responses to experimentally-evoked pain between obese and non-obese human participants post-puberty (i.e. >16 years) were sought. Eligible studies published between January 1950 and May 2017 were identified by searching OVID, MEDLINE, EMBASE and Science Direct.
Explanation:
Methodological quality of included studies was assessed using the ‘QualSyst’ questionnaire. Of 1106 references identified only nine studies (683 participants) were eligible for review. Pressure pain was assessed in five studies and electrical pain in three studies. Two studies investigated thermal pain. Obesity was categorized according to body mass index (BMI) or as weight as a percentage of ideal body weight. Six of the nine included studies were of low methodological quality. There was a lack of extractable data to pool for meta-analysis of studies using thermal or electrical pain. A forest plot of data extracted from four studies on pressure pain threshold found no differences between obese and non-obese groups (overall effect size was Z=0.57, p=0.57).
Conclusion:
Small sample size was the main limitation in all studies. Participants with obesity were more sensitive to mechanical noxious stimuli than non-obese participants in three of five studies. However, overall, it was not possible to determine whether there are differences in pain sensitivity response to experimental stimuli between obese and non-obese individuals.
1. INTRODUCTION
Obesity and chronic pain are global public health problems [1, 2]. Evidence suggests that individuals who are obese are more likely to report chronic musculoskeletal pain [3-5] including fibromyalgia [6], low back pain [7], and osteoarthritis [8]. It has been claimed that pain is mediated in part by excess load causing stress on weight bearing joints leading to potential tissue damage [9, 10], although mechanisms other than mechanical overloading are likely to be involved because increased pain occurs in both weight bearing and non-weight bearing parts of the body.
Recently, attention has focussed on systemic inflammation associated with central obesity and whether this influences pain [11, 12]. Individuals with central obesity have a propensity for systemic inflammation resulting in body tissue that is swollen and sensitive to stimuli with the potential to produce pain [5, 13]. The excessive visceral fat associated with central obesity contributes to an increase in circulating levels of pro-inflammatory markers (e.g. prostaglandins, C-reactive protein and cytokines such as interleukins (Interleukin-6), tumour necrosis factor alpha (TNF-alpha) and leptin) and decreased levels of anti-inflammatory markers (e.g. adiponectin). Pro-inflammatory markers sensitise nociceptors in peripheral tissue lowering their thresholds of activation (peripheral sensitisation) and amplifying peripheral nociceptive activity. This sensitises central nociceptive transmission cells (central sensitisation) resulting in increased pain [14]. However, the relationship between central obesity, systemic inflammation and pain remains unclear. An epidemiological study investigated the association between obesity and pain prevalence among older adults in 407 elderly individuals and found that central obesity had the strongest independent association with pain, although the relationship was not explained by markers of inflammation or insulin resistance [15].
Psychophysical methods can be used to quantify pain by measuring and individual’s response to noxious stimuli (i.e. experimentally-evoked pain). Common measures include pain threshold, the magnitude of a stimulus necessary to evoke the first sensation of pain, and pain tolerance, the magnitude of a stimulus necessary to evoke pain that an individual can no longer endure. Pain threshold and pain tolerance decrease in the presence of systemic inflammation [16, 17]. The aim of this systematic review was to determine whether response to experimentally-evoked pain differed between obese and non-obese individuals.
2. METHODS
2.1. Data Sources and Search Methods for Identification of Studies
This review used the guidelines for Preferred Reporting Items for Systematic Reviews (PRISMA) [18]. Studies published between January 1950 and May 2017 were identified by searching OVID, MEDLINE, EMBASE and Science Direct using the following keywords and MeSH terms: [pain sensitivity OR experimental pain OR pain measurement OR quantitative sensory testing OR pressure pain thresholds OR heat pain thresholds] AND [obesity OR overweight OR BMI]. We also searched manually for relevant studies in the reference list of hey articles.
2.2. Criteria for Considering Studies for this Review
Studies were included if they:
- used healthy human participants post puberty (i.e.>16 years);
- measured response to experimentally-evoked pain (e.g. pain threshold, pain tolerance, pain intensity or unpleasantness rating);
- compared obese and non-obese participants by measuring body mass index (BMI, e.g. weight and height) and/or aspects of body fat (e.g. percentage body fat) by skin fold thickness, waist-hip ratio or dual-energy X-ray absorptiometry (DXA) and;
- were written in any language with non-English reports translated if necessary.
Studies were excluded if they involved a treatment intervention.
2.3. Study Selection
Two reviewers (RA, OAT) screened titles and abstracts independently to identify potentially relevant studies. Reviewers were not blind to the names, institution, journal, or study results. Full reports of potentially relevant studies were obtained and screened for inclusion by RA and OAT. Disagreements were resolved by consensus with the whole review team with MIJ acting as arbiter.
2.4. Data Extraction
Data was extracted from included studies by RA and OAT independently including number of participants, age and gender, measurements to categorise individuals into groups (e.g. non-obese and obese), experimental pain technique (e.g. noxious stimuli) and pain outcome measures. Agreement between the two reviewers was tested and reported.
2.5. Assessment of Study Methodology, Outcome and Heterogeneity
The methodological quality of the included studies was assessed independently by two reviewers (RA, OAT) using 11 criteria developed by Kmet et al. [19]. The assessment criteria of the quality of the research were based on the following questions:
- Was the research question/objective sufficiently described?
- Was the study design evident and appropriate?
- Was the method of participant/comparison group selection or source of information/input variables described and appropriate?
- Were participant and comparison group, (if applicable), characteristics sufficiently described?
- Was the outcome and, (if applicable), exposure measure(s) well defined and robust to measurement/misclassification bias?
- Were means of assessment reported?
- Was the sample size appropriate?
- Were the analytical methods described/justified and appropriate?
- Was some estimate of variance reported for the main results?
- Was there any control for confounding factors? Was there sufficient detail in the reporting of results?
- Did the results support the conclusion?
It was planned that quantitative data for outcomes would be pooled where possible. In addition, the standardised mean difference between obese and non-obese participants with 95% CI (continuous data) or relative risk (dichotomous data) would be calculated. It was also planned that heterogeneity between comparable studies would be assessed, using Review Manager (RevMan) [Computer program], Version 5.3. (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). A tally of outcomes was planned based on at least one statistically significant difference between groups at any time point if it was not possible to pool data.
3. RESULTS
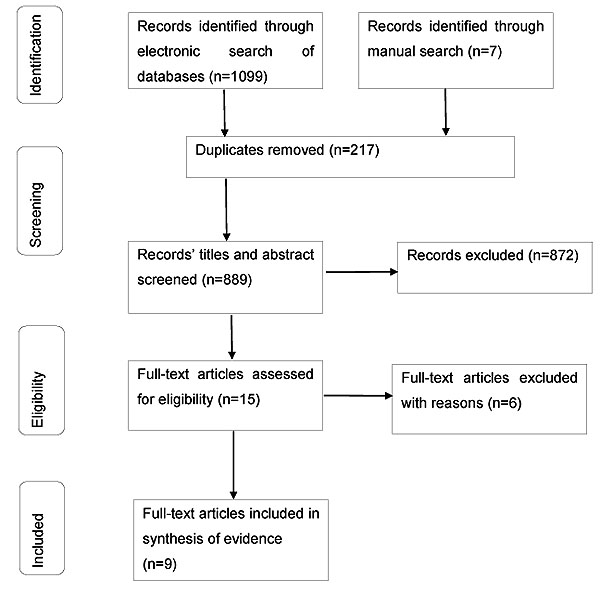
There were 1099 references identified in the literature search of which 217 were duplicates (Fig. 1). An additional seven references were identified through manual searches of reviews and discussion articles retrieved from the original search. This produced 889 references for screening through titles and abstracts. Of these, 872 references were excluded because they were either not relevant or were studies that had been conducted on animals or individuals with pre-existing pain conditions (i.e. clinical pain). The full reports of 15 potential studies were assessed for eligibility and eight of these were excluded (Table 1).
| Reference | Author, Year | Reason for Exclusion |
|---|---|---|
| [17] | (Benson, et al. 2012) | No attempt was made to categorise the sample into obese and non-obese by measuring BMI or body fat |
| [20] | (Eberle, et al. 2010) | No attempt was made to categorise the sample into obese and non-obese by measuring BMI or body fat |
| [21] | (Euteneuer, et al. 2011) | The study involved patients and control groups. No attempt was made to categorise healthy participants control into obese and non-obese according to BMI or body fat |
| [22] | (Manson, 2010) | Discussion article |
| [23] | (Tashani. et al. 2010) | All participants were normal BMI (i.e. not obese) |
| [24] | (Price, et al. 2013) | Healthy participants received a placebo intervention as part of a randomised controlled clinical trial |
3.1. Characteristics of Included Studies
Nine studies (683 participants) were included for review Table 2a. Five of these studies were conducted before the year 2000. All reports were written in English language. Study sample size varied between 38 and 206, and age varied between 16 and 84 years. Three studies used only females [25-27] and six studies recruited males and females. Obesity was categorized according to BMI [27, 28, 29, 30] or actual weight as a percentage of ideal body weight [31] or other weight measures. Only one study measured body fat directly using dual-energy X-ray absorptiometry (DXA) although participants were categorised according to BMI [29].
| Study | Study Participants [Sample size (M/F; age)] | Classification of Obesity | Noxious Stimuli | Outcome Measure |
Data Obese [Mean+SD] |
Data Non-Obese [Mean+SD] |
Difference P value |
Authors’ Conclusion |
Methodological Quality Score (%) Reviewers’ Comment |
|---|---|---|---|---|---|---|---|---|---|
| McKendall and Haier (1982) [31] | N = 60(28/32; 20-67 years) N obese = 26(8/18) N non-obese = 34(20/14) |
Obese >130% ideal body weight Non-obese <130% ideal body weight |
Constant blunt pressure applied at 3lbs applied to finger using a “pressure bearing device” | Pain Threshold | 59.4+48.15s |
95.51+53.36s |
<0.001 |
Obese more sensitive to noxious stimuli than non-obese | 55% |
| Pain Tolerance | 81.88+56.09s | 131.03+ 51.14s | <0.01 | ||||||
| Raymond et al. (1995) [27] |
N = 104(0/104; 19-50 years) N obese without binge eating disorder [non-BED] = 33(0/33) N obese with binge eating disorder [BED] = 27(027) N non-obese [control] = 44(0/44) |
BMI | Blunt pressure applied at 64gs-1 to finger using an Ugo Basile analgesia meter | Pain Threshold |
non-BED= 384+24g BED =453+31g |
356+19g [control] |
0.38 |
No difference in sensitivity to noxious stimuli between obese and non-obese individuals | 48% No statistically significant correlation coefficient (r) between BMI and pain threshold or tolerance |
| Pain Tolerance | non-BED =544+32g BED=612+42g |
552+29g [control] | 0.297 | ||||||
| Khimich (1997) [32] | N = 206 (114/92;18-84 years) N obese (stage II-III) = Not reported N overweight (stage I obese) = Not reported N ‘non-obese’=Not reported |
Used Broca index [details not reported] into Obese (stage II-III) Obese stage I (overweight) Normal |
Needle (sharp) pressure applied to the forearm | Expression of mild pain [assumed to be pain threshold] Expression of severe pain [assumed to be pain tolerance] |
72.9g [SD not reported] |
Normal = 40.5g [SD not reported] Overweight = 57.6g [SD not reported] |
Not reported |
Obese less sensitive to noxious stimuli than non-obese | 21% Minimal detail provided in report Overweight measure for pain tolerance reported as 91.3g in Abstract and 91.8g in Results |
| 116.2g [SD not reported] | Normal = 76.5g [SD not reported] Overweight=91.8g [SD not reported] |
Not reported | |||||||
| Astita et al. (2015) [29] | 38 adults (18 women) were grouped as normal weight(n=22) or obese (n=16) | BMI | Somedic Algometer applied at thenar eminence | Pain Threshold | 340.93±93.58kPa | 447.45±203.72kPa | 0.039 | Obese more sensitive to blunt pressure stimuli than non-obese | 73% |
| Tashani et al. (2017) [30] | 72 healthy participants (37 women) divided into 3 groups according to their BMI: Normal=25 Overweight= 24 Obese= 25 |
BMI values were: Obese= 34.4+3.90 kg/m2 Normal =22.1+2.04 kg/m2 |
Somedic Algometer applied at thenar eminence |
Pain Threshold | 620.72+423.81kPa | 1154.70+847.18kPa | 0.005 | Obese more sensitive to blunt pressure stimuli than non-obese | 88% |
There were five studies that measured response to pain evoked by noxious pressure. Three studies [31, 29, 30] found that blunt pressure pain threshold was lower in obese participants than non-obese, whereas Raymond et al. [27] found that there were no statistically significant differences in pain threshold or pain tolerance in obese participants compared with non-obese. Khmich [32] claimed that expression of mild pain (which we have assumed to be pain threshold) and severe pain (which we have assumed to be pain tolerance) to pin-prick was higher in obese participants than non-obese but there was no statistical analysis to support the claim.
Pradalier et al. [33] used percutaneous electrical stimulation of the sural nerve to evoke nociceptive-flexion reflexes and found that the threshold to elicit the reflex was lower in obese compared with non-obese individuals. They also found an inverse correlation between percentage of ideal body weight and the threshold of nociceptive reflex. However, the nociceptive-flexion reflex of the sural nerve is not a measurement of pain threshold nor tolerance so this study was not selected for further consideration in our review. Nonetheless, it was interesting that the threshold for the reflex response was dependent on the amount subcutaneous fat tissue. There were three studies that measured response to pain evoked by electrical stimuli applied transcutaneously [25, 26, 28]. Zahorska-Markiewicz et al. [25] found that pain threshold to an electrical stimulus on the forearm and arm was higher in obese participants compared with non-obese (effect size=0.61 at the forearm, 0.53 at the arm). The same investigators conducted a similar study to investigate the relationship between circadian rhythms and electrical pain threshold during weight-reducing treatment and found that obese participants had higher pain thresholds compared with non-obese at baseline wit the effect size = 0.53 [26]. Maffiuletti et al. [28] found that there were no significant differences in intensity of pain associated with the ‘highest tolerated threshold’ of transcutaneous electrical neuromuscular stimulation of the quadriceps between male obese and non-obese participants and female obese and non-obese participants when analysed separately. However, they did not report the analysis of obese and non-obese participants when males and females were combined.
Two studies found that heat pain threshold values were higher in obese participants compared with non-obese individuals [29, 34, 35] (Table 2b), although the study by Miscio et al. [34] found that obese individuals are more sensitive to cold stimuli than non-obese (effect size for index finger=-0.43, and for little finger=-0.35).
| Study | Study Participants [Sample size (M/F; age)] | Classification of Obesity | Noxious Stimuli | Outcome Measure |
Data Obese [Mean+SD] |
Data Non-Obese [Mean+SD] |
Difference P value |
Authors’ Conclusion |
Methodological Quality Score (%) Reviewers’ Comment |
|---|---|---|---|---|---|---|---|---|---|
| Zahorska-Markiewicz et al. (1983) [25] | N = 48(0/48; 16-52 years) N obese = 20 (0/20) N non-obese = 20 (0/20) N underweight = 8(0/8) |
Percentage excess against ideal body weight from Metropolitan Life Insurance Tables Mean % excess obese = 87.9%, non-obese = 1.6% and underweight = -10.1% |
Transcutaneous electrical stimulation of forearm with amplitude increasing from 0-18mA | Pain Threshold | Forearm = 13.03+3.27mA Arm = 8.83+4.31mA |
Forearm = 7.55+3.76mA Arm = 5.56+2.32mA Forearm [underweight] = 5.88+3.18mA Arm [underweight] = 6.1+5.34mA |
Forearm <0.001 Arm P=<0.01 |
Obese less sensitive to noxious stimuli than non-obese | 32% Positive correlation between pain threshold and body weight, and weight excess |
| Zahorska-Markiewicz et al. (1988) [26] | N = 49(0/49; 24-61 years) N obese = 35(0/35) N non-obese = 14(0/14) |
Percentage excess against ideal body weight from Metropolitan Life Insurance Tables Mean % excess obese = 64.5%, non-obese = not reported |
Transcutaneous electrical stimulation of forearm with amplitude increasing from 0-20mAmin-1 at various time points throughout the day and over a 4 week weight reducing treatment phase |
Pain Threshold [1st measure taken at 7am pre-treatment] | 12.5+4.4mA | 7.3+3.8mA | <0.001 | Obese less sensitive to noxious stimuli than non-obese | 36% Main focus of study was to investigate relationship between circadian rhythms and electrical pain threshold during weight-reducing treatment |
| Maffiuletti et al. (2011) [28] | N = 67 (34/33) N obese = 32(M/F not reported) N ‘non-obese’ =35(M/F not reported) |
BMI Obese >30kg/m2 |
Transcutaneous electrical stimulation of quadriceps muscle to elicit muscle contraction | Pain Intensity (100mm VAS) of ‘highest tolerated threshold’ to elicit contraction | M=46+27mm F=56+26mm |
M=43+26mm F=48+27mm |
Not significantly different” [P value not reported] | No difference in sensitivity to noxious stimuli between obese and non-obese individuals | 66% Conclusion “Current tolerance to motor stimulation was reduced in obese individuals … whereas pain was not influenced by gender or obesity” |
3.2. Assessment of Study Methodological Quality
The two reviewers (RA, OAT) assigned identical total study methodological quality scores for six of the nine studies. After discussion the consensus between the team was to take the average of the scores for each component of the assessment of the remaining three studies where there was a difference in methodological quality scoring. Six studies scored more than 50% in the quality assessment criteria. Three studies were classified as poor methodological quality.
3.3. Assessment of Study Outcome and Heterogeneity
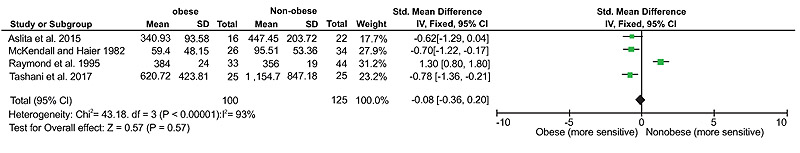
It was possible to pool data from four of the five studies that recorded pressure pain threshold to calculate standardized mean difference (SDM) based on a fixed effects model. There were no statistically significant differences between obese and non-obese groups in blunt pressure pain threshold (SMD (95% CI) = -0.08 (-0.36 to 0.20), overall effect size Z = 0.57, p = 0.57, (Fig. 2). There was substantial heterogeneity between the studies with I2 calculated to be 93%.
It was not possible to pool data from the three studies that used electrical stimuli because two measured pain threshold and one measured pain intensity. The authors of two of these studies (both from the same investigating team) concluded that obese individuals were less sensitive to painful stimuli than non-obese individuals [25, 26]. The authors of the third study concluded that there were no differences in pain intensity [28]. It was not possible to pool data from thermal stimuli because there were only two studies. The authors of these studies concluded that obese participants were less sensitive to noxious thermal stimuli [34, 30].
4. DISCUSSION
Nine studies were included in this systematic review but findings were inconsistent and methodological quality of the studies low. The meta-analysis of four studies on blunt pressure pain threshold suggested that there were no differences between obese and non-obese individuals. Data from two studies on heat pain indicated that obese individuals were less sensitive to noxious heat stimuli than non-obese individuals. It was noteworthy that six of the nine included studies were published over 15 years ago, and the influence of confounding variables including gender, age, comorbidities (e.g. hypertension, diabetes), and socio-economic factors were not assessed. Thus, there is insufficient high quality research to determine with any degree of confidence whether there are differences in sensitivity to experimentally-evoked pain between obese and non-obese individuals.
A secondary analysis of data obtained as part of a double-blind, placebo controlled, cross-over clinical trial that investigated the efficacy of naltrexone by Price et al. [24] was excluded from this review because we were unable to obtain the report of the original study. Data used in the secondary analysis was “pertaining to the placebo session”. We interpreted this to mean that data collected from the placebo arm of the clinical trial was used to compare pain sensitivity between obese and non-obese individuals. This secondary analysis by Price et al. was not eligible for inclusion in our systematic review because it involved a placebo intervention which we specified as an exclusion criterion and no details were provided about the procedures used to administer the placebo intervention. Nonetheless, this secondary analysis is the most comprehensive analysis of pain sensitivity responses in obese individuals to date. The secondary analysis found no significant differences in pressure pain threshold on the thenar eminence and thumbnail. Inclusion of this data in our review would not have altered our conclusion or decision not to pool data for meta-analysis.
Interestingly, Price et al. [27] found that contact heat pain threshold and contact cold pain threshold was higher in obese compared with non-obese participants (i.e. lower pain sensitivity) when measured on at the abdomen but not at the forehead or thenar eminence. These differences correlated with measures of adiposity and the amount of excess subcutaneous fat. Price et al. also found that obese individuals had higher pressure pain threshold (i.e. were less sensitive) than non-obese individuals suggesting inconsistency with the findings of studies included in our review. This may be due to variations in the amount of adipose tissue at different body sites [36]. Unfortunately, Price et al. did not measure pressure pain threshold over the abdomen. Price et al. [24] suggested that local mechanical and chemical factors played a role in the observation of differences in pain thresholds at different body sites and that generalized factors such as systemic inflammation leading to changes in centralized pain processing were less likely to be involved (Table 2c). They suggested that localised reductions in sensitivity to noxious thermal stimuli may be due to stretching of the skin due to excess fat and that this decreased the density of epidermal nerve fibres associated with cold detection and heat pain (A-delta fibres) and warm detection, heat pain and cold pain (C-fibres).
| Study | Study Participants [Sample size (M/F; age)] | Classification of Obesity | Noxious Stimuli | Outcome Measure |
Data Obese [Mean+SD] |
Data Non-Obese [Mean+SD] |
Difference P value |
Authors’ Conclusion | Methodological Quality Score (%) |
|---|---|---|---|---|---|---|---|---|---|
| Miscio et al. (2005) [34] | 21 obese non-diabetic volunteers versus 20 non-obese healthy volunteers | BMI Obese >30kg/m2 |
A computerized quantitative device (Medoc Ltd; TSA II-2001) was used to test the thresholds for heat and cold-evoked pain (HP-CP) | Cold Pain on index finger | 12.8+10.2oC | 20.5+5.1oC | 0.004 | Obese higher heat pain threshold and lower cold pain threshold than non-obese | 72% |
| Heat Pain on index finger | 46.4+3.6oC | 42.6+3.6oC | 0.002 | ||||||
| Cold Pain on little finger | 13.1+10.1oC | 19.3+6.0oC | 0.021 | ||||||
| Heat Pain on little finger | 46.2+3.9oC | 43.3+3.5oC | 0.016 | ||||||
| Tashani et al. (2017) [30] | 74 healthy participants (37 women) divided into 3 groups according to their BMI: Normal=25 Overweight= 24 Obese= 25 |
BMI values were: Obese=34.4+3.90kg/m2 Normal=22.1+2.04kg/m2 |
A computerized quantitative device (Medoc Ltd; TSA II-2001) was used to test the thresholds for heat-evoked pain threshold and tolerance | Heat Pain Threshold at thenar eminence | 42.29+3.4oC | 42.75+3.5oC | 0.26 | No differences between groups | 88% |
| Heat Pain Tolerance at thenar eminence | 48.33+2.61oC | 48.23+2.1oC | 0.59 | ||||||
| Heat Pain Threshold at the waist | 41.38+3.07oC | 41.39+3.07oC | 0.9 | ||||||
| Heat Pain Tolerance at the waist | 46.71+1.77oC | 47.35+1.6oC | 0.18 |
In contrast, other research suggests that the pro-inflammatory state observed in individuals with obesity has been attributed to the ratio of circulating pro-inflammatory: anti-inflammatory cytokines mediated by visceral fat adipose cells and leading to alterations in pain sensitivity response. It is not known whether the local adipose tissue environment is more abundant in, and thus prone to be affected by, anti-inflammatory cytokines whereas the systemic circulatory environment is more abundant in pro-inflammatory cytokines. Clearly, there is a need for further research to determine whether changes in pain sensitivity are a generalised phenomenon regardless of body location tested, perhaps mediated by changes in centralised pain processing, or whether changes are localised, reflecting differences in fat at various body sites. Moreover, studies included in our review did not measure cytokines and other substances (such as leptin) that are known to influence nociceptor sensitivity.
One limitation of this systematic review was that the evidence synthesis was based on a low number of studies and some of these studies where published before recent development in technologies and methodologies to assess pain sensitivity response. The studies included in our review were vulnerable to high risk of bias including failure to blind investigators from group identity. We excluded studies measuring pain sensitivity in individuals with a pre-existing pain condition because it would have increased heterogeneity of the sample populations and further complicated the analysis of findings.
What can be learned from this systematic review findings is that to design a new study to investigate the differences between obese and non-obese individuals certain conditions have to be met:
- Use of a better obesity indicator than BMI as BMI is a crude categorization of obesity and can be misleading in judging obesity of some sub-population (e.g. athletes). There are much better methods in categorization of obesity including measure of total body fat.
- Data provided in this systematic review should be used to calculate the appropriate sample size of participants to achieve sufficient statistical power to reach clinical meaningful differences.
- Standardised pain induction techniques including Quantitative Sensory Testing should be used in future studies.
CONCLUSION
It was not possible to determine whether there are differences in pain sensitivity between obese and non-obese individuals because there were too few studies and those that exist were contradictory and had low methodological quality ratings. Study designs varied considerably with marked differences in the type of evoking-stimuli used and body sites tested. Concentrations of pro-inflammatory and anti-inflammatory mediators were rarely measured. There is a need for studies that measure pain sensitivity at different body sites that assess body-fat distribution and measure systemic markers of inflammation.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
All authors declare that there are no conflicts of interest relevant to this study.
ACKNOWLEDGEMENTS
The Higher Institute of Medical professions, Al-Bayda, Libya, for financial support to undertake this study. An abstract of this paper was presented at the British Pain Society Annual Scientific Meeting in Aril 2014 as a poster presentation with interim findings.